
Master the supplement effectiveness workflow for health
Share
TL;DR:
- Most supplement disappointments stem from flawed selection processes rather than personal failure, especially for adults over 40. Applying a structured, evidence-based workflow helps differentiate between supplement efficacy and product quality, ensuring informed choices grounded in scientific research. Personalization, rigorous evidence assessment, and safety screening are essential for achieving meaningful results in healthy ageing.
You have likely spent real money on supplements that seemed promising, followed instructions carefully, and still felt no different after months of consistent use. This experience is common among adults over 40, and it rarely comes down to a simple lack of willpower or compliance. The truth is that most supplement disappointments trace back to a flawed selection process, not a flawed person. This article walks you through a structured, science-supported workflow for evaluating supplement effectiveness, so that every choice you make is grounded in evidence rather than packaging claims.
Table of Contents
- Why supplement effectiveness requires a workflow
- What you need before you start: Preparation checklist
- Step-by-step: The supplement effectiveness workflow
- Common pitfalls and how to ensure real-world results
- Why most ‘silver bullet’ supplement strategies fail: Lessons from real-world workflows
- Explore evidence-based supplement solutions with Vivetus
- Frequently asked questions
Key Takeaways
| Point | Details |
|---|---|
| Separate evidence from claims | Distinguish between what works in clinical studies and what a product actually delivers. |
| Follow a stepwise workflow | Systematic steps ensure you choose supplements with real potential for results. |
| Always check product integrity | Quality, purity, and correct dosing are essential for effectiveness and safety. |
| Assess for your needs | Consider your health status and goals, not just general claims. |
| Prioritise ongoing verification | Regularly monitor results and adapt your supplement strategy based on outcomes. |
Why supplement effectiveness requires a workflow
The gap between what supplement research shows and what ends up inside a product is wider than most people realise. A study might demonstrate genuine benefits for a specific ingredient at a precise dose, in a particular molecular form, with a defined population. The commercial product sitting on a shelf is under no legal obligation to replicate any of those conditions exactly. This mismatch is where most supplement disappointments begin.
Evaluating supplement benefits effectively requires separating two distinct questions: does the ingredient work in principle, and does this specific product deliver it in the form, dose, and quality the evidence actually supports? These are not the same question, and treating them as identical is the single most common error adults make when selecting supplements.
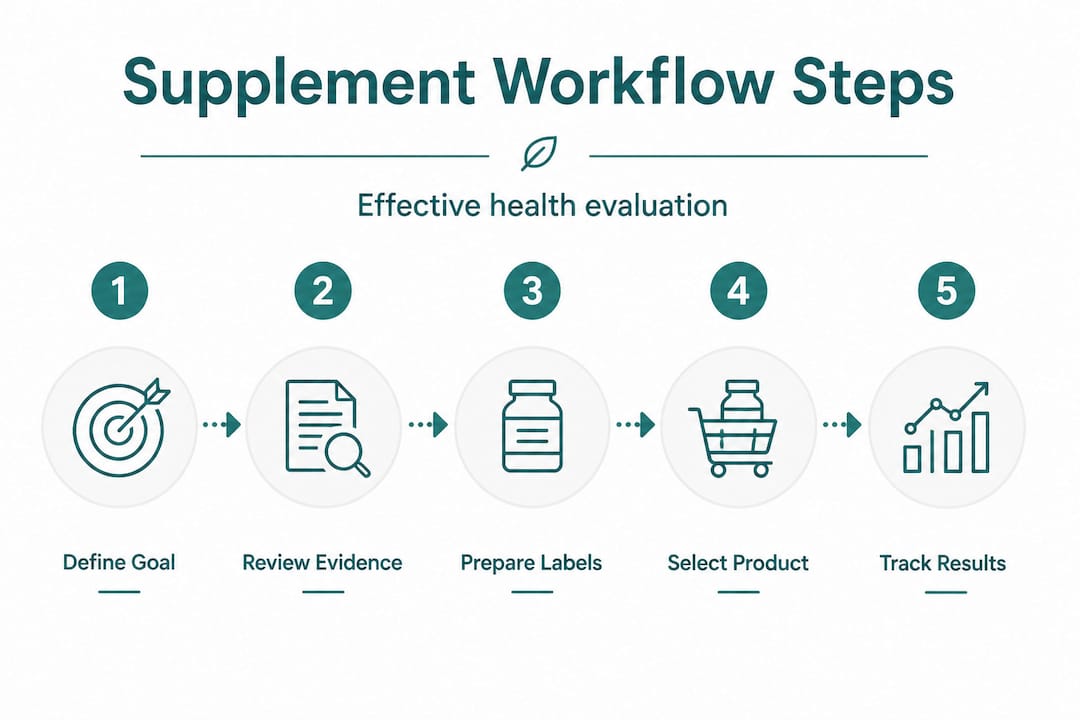
A structured workflow addresses this directly. As one framework puts it, a robust effectiveness workflow separates efficacy evidence for an ingredient or form from execution quality at the product level. Without that separation, you might take a well-studied ingredient every day and absorb almost none of it because the manufacturer chose a cheaper, poorly bioavailable form.
Three pillars support any sound supplement evaluation:
- Evidence for effect: Does credible research show this ingredient produces the outcome you want, at a dose you can realistically take?
- Execution quality: Does the product contain the right ingredient form, at the studied dose, with verified purity and batch integrity?
- Safety profile: Are there known interactions, contraindications, or upper limits relevant to your health history?
Research institutions increasingly recognise that supplement effectiveness studies require rigorous, reproducible methods and transparent reporting, including product integrity characterisation. Without this standard, even well-intentioned research can fail to translate into reliable consumer guidance.
What you need before you start: Preparation checklist
Before applying any evaluation workflow, preparation matters. Walking into the process without a clearly defined health goal is one of the fastest routes to wasted effort. Your preparation phase defines the boundaries of what you are assessing and prevents you from being distracted by marketing claims that address outcomes irrelevant to your actual needs.
Start by writing down your specific health outcome. Not “I want to feel healthier,” but rather something precise: maintaining bone density, supporting cardiovascular function, improving sleep quality, or sustaining cognitive performance as you age. This precision shapes every step that follows.
Gather the following before you begin:
- Your current supplement and medication list, including doses and timing
- Any relevant lab results or clinical notes from the past 12 months
- Your health history, particularly conditions that affect absorption or metabolism
- The ingredient panels of any products you are currently considering, ideally photographed for easy reference
- Batch or lot numbers for any products you plan to assess for quality verification
Understanding how to interpret a label is also necessary. Supplement labelling can be deliberately ambiguous, using proprietary blends or vague ingredient names that make it difficult to verify whether the studied form and dose are present. Knowing what to look for before you start saves significant time.
It is also worth noting that analytical method validation is a necessary step in supplement research workflows because complex natural product matrices can vary significantly between batches and manufacturers. This means that even a product with an accurate label today may differ from the same product purchased six months later.
| Preparation item | Why it matters |
|---|---|
| Specific health outcome | Focuses your evidence search |
| Current supplement/medication list | Enables interaction screening |
| Lab results and clinical notes | Identifies baseline deficiencies or conditions |
| Ingredient panel photographs | Allows systematic label comparison |
| Batch and lot numbers | Supports quality and recall verification |
Pro Tip: Keep a dedicated folder on your phone with photos of every supplement label and its batch number. If a product is reformulated or recalled, you will have the exact reference needed to act quickly.

Step-by-step: The supplement effectiveness workflow
With preparation complete, the structured assessment process can begin. This workflow applies to any supplement category, from omega-3 fatty acids to magnesium glycinate to coenzyme Q10.
-
Define the specific health outcome. State clearly what result you are seeking, the timeframe, and how you will measure it. For example: “I want to assess whether magnesium supplementation improves my sleep onset time, measured by a sleep tracker over eight weeks.”
-
Identify the ingredient, dose, and form studied. Not all forms of a nutrient behave identically in the body. Magnesium oxide and magnesium glycinate, for instance, have very different absorption rates. Identify precisely which form and dose the research used.
-
Search for the best available evidence. Prioritise the evidence hierarchy: systematic reviews and meta-analyses sit at the top, followed by randomised controlled trials, then observational studies, then animal or cell-based data. Expert opinion and traditional use sit at the bottom. The higher on that hierarchy your evidence sits, the more confidence you can place in the finding.
-
Assess evidence quality, not just existence. A published study is not automatically strong evidence. Look for GRADE certainty ratings, effect sizes, and risk-of-bias assessments rather than just a p-value. Systematic reviews assessing supplement effectiveness use PRISMA and PROSPERO registration alongside risk-of-bias tools to communicate confidence levels. A small, unregistered trial with a high risk of bias tells you very little.
-
Compare the product label against the evidence. Does the product contain the exact ingredient form studied? Is the dose within the range shown to be effective? Is the product independently verified for purity and label accuracy? The evidence for supplement choices rarely transfers automatically from a study ingredient to any commercial product.
-
Screen for safety and interactions. Discuss your shortlist with a qualified clinician or pharmacist, particularly if you take prescription medications. Some supplements interact with common medicines in ways that are not obvious from the label.
-
Track results and iterate. Set objective outcome measures before you start. Reassess after a defined period, typically eight to twelve weeks for most compounds. If results are absent, consider whether the dose, form, or product quality needs reassessing before concluding the ingredient does not work for you.
| Workflow step | Common shortcut/mistake |
|---|---|
| Define outcome | Choosing a supplement before defining the goal |
| Identify ingredient and form | Assuming all forms of a nutrient are equivalent |
| Search for evidence | Accepting brand-cited studies without checking quality |
| Assess evidence quality | Treating any published study as strong evidence |
| Compare product to evidence | Assuming a recognised brand name guarantees studied dose |
| Safety screening | Skipping clinician review when taking medications |
| Track and iterate | Stopping too early or changing multiple variables at once |
The NIH Office of Dietary Supplements outlines a practical consumer workflow that aligns closely with this sequence: define the goal, identify the active ingredient, assess the best available evidence, verify product execution, and check safety. These are not optional steps. Skipping any one of them introduces meaningful uncertainty into your decision.
Pro Tip: When reading a study cited by a supplement brand, check whether the study used the exact same product or a researcher-prepared formulation. Many positive trials use pharmaceutical-grade compounds that bear little resemblance to the commercial product being sold.
The distinction between evidence-backed longevity supplements and those built on marketing alone becomes clear when you apply this step-by-step process. The evidence hierarchy does the sorting for you.
Common pitfalls and how to ensure real-world results
Even with a solid workflow in place, several real-world factors consistently derail supplement effectiveness for adults over 40. Recognising them in advance protects both your health and your investment.
One of the most significant issues is subgroup specificity. A supplement may show genuine benefits in studies conducted with individuals who have a baseline deficiency or a specific health condition. If you do not share that baseline, the same ingredient may offer little benefit regardless of dose or quality. Supplement effectiveness claims are frequently supported for specific subgroups or outcomes, and your workflow must include this edge-case logic.
The regulatory context in most countries adds another layer of risk. The FDA does not evaluate supplement safety or effectiveness before products reach the market. This places the entire burden of quality verification on the consumer and the retailer. It is not a flaw in any single product but a structural feature of the industry that demands informed navigation.
Watch for these specific pitfalls:
- Underdosing: Many commercial products contain doses well below those used in clinical studies, sometimes to reduce cost or fit a standard capsule size.
- Wrong form: The studied form of an ingredient may be more expensive to source, leading manufacturers to substitute a cheaper, less bioavailable alternative with a similar-sounding name.
- Unrecognised interactions: Certain supplements, including vitamin K, St John’s Wort, and high-dose fish oil, interact with common prescription medications in clinically significant ways.
- Absence of result tracking: Without baseline measurements and a defined follow-up period, it is genuinely impossible to know whether a supplement is working.
- Batch variation: Natural product supplements can vary between production runs; always note the batch number when starting a new supply.
“Because supplement claims may be supported for specific subgroups or outcomes, the evaluation process should always incorporate edge-case logic, considering deficiency status, disease stage, and outcome specificity.” This principle applies directly to adults over 40, where individual variation in absorption, metabolism, and baseline nutritional status tends to increase with age.
Review supplement safety tips as part of your regular practice, and consult a nutritional supplements guide to address specific edge-case considerations relevant to healthy ageing.
Why most ‘silver bullet’ supplement strategies fail: Lessons from real-world workflows
The supplement industry generates billions of pounds annually, and a meaningful portion of that revenue comes from adults who have been disappointed before but remain hopeful that the next product will finally work. That hope is understandable. The discipline required to resist it is harder than most people expect.
From working within the evidence-based nutrition space, a pattern becomes clear: the people who achieve consistent, measurable benefits from supplementation are not the ones who follow trends or respond to urgency marketing. They are the ones who apply a repeatable process, adjust based on objective results, and accept that no single ingredient solves a complex biology.
The uncomfortable reality is that bioavailability, individual metabolism, gut microbiome composition, and baseline nutrient status vary enough between people that the same product will produce different outcomes in different individuals, even when the evidence base for the ingredient is strong. This is not a reason to abandon supplementation. It is a reason to personalise your approach and stop expecting the results reported in studies conducted on populations very different from you.
Genuine effectiveness in healthy ageing requires iteration. You define a goal, gather evidence, select a quality product, track your response, and adjust. This process is less exciting than discovering a “breakthrough” ingredient, but it is the only approach that reliably separates what works for you from what merely works in theory. A well-structured guide to supplement use reflects this iterative mindset, providing a framework that grows more accurate as you apply it consistently.
The most common reason supplementation strategies fail is not a lack of information. It is the absence of process discipline. With the workflow described in this article, you have the structure needed to move from hopeful trial-and-error to evidence-guided practice.
Explore evidence-based supplement solutions with Vivetus
Applying this workflow becomes significantly more practical when you have access to reliable resources and curated product information in one place.
Vivetus is built around the same evidence-first principles covered in this article, providing a platform where supplement choices are supported by scientific context rather than marketing language. Whether you are reviewing the evidence for a specific ingredient, comparing product quality indicators, or looking for practical guidance on safe supplementation for healthy ageing, Vivetus offers structured resources to support each stage of your workflow. Explore curated supplement information and supplement safety steps to take the next informed step in your longevity-focused supplement practice.
Frequently asked questions
How can I tell if a supplement is supported by strong evidence?
Look for systematic reviews or randomised controlled trials, as these provide the highest quality evidence for supplement effectiveness. The evidence hierarchy places systematic reviews and meta-analyses above all other study types.
What should I check on a supplement label for effectiveness?
Ensure the product lists the clinically studied ingredient, dose, and form, and check for independent quality verification where possible. Product integrity characterisation includes identity, purity, composition, dosage, and stability, all of which should be verifiable.
Are supplements for healthy ageing pre-approved like medicines?
No, supplements are not pre-approved for safety or effectiveness. The FDA does not review products before they reach the market, so consulting your healthcare provider before use is essential.
How do I avoid wasting money on ineffective supplements?
Follow a comparative, evidence-prioritised workflow rather than relying on marketing claims or price alone. Efficacy evidence for an ingredient does not automatically transfer to a commercial product due to differences in bioavailability and manufacturing execution.
What does ‘edge-case logic’ mean in supplement effectiveness?
It means accounting for specific personal factors, such as your baseline deficiency status, health stage, and the outcomes that are genuinely relevant to you. Workflow edge-case logic ensures that population-level findings are tested against your individual context before you commit to a supplement.
